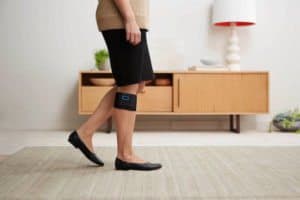
NeuroMetrix, Inc. (Nasdaq: NURO) reported it has started shipping Quell wearable pain relief devices out of its production and fulfillment facility in Woburn, MA. The initial shipments are directed to leading doctors and thought leaders in chronic pain. The company anticipates that it will start fulfilling orders in late May that were placed through its pre-order campaign on the Indiegogo. The company expects to clear the remaining backorders and begin ongoing commercial shipments for new orders in June. The Company has an order backlog of about 2000 Quell devices.
 Quell is an over-the-counter wearable pain relief device that utilizes proprietary non-invasive neurostimulation technology to provide pain relief. The device is designed for people with conditions such as diabetic nerve pain, low back and leg pain, fibromyalgia, and pain associated with osteoarthritis. The advanced wearable device is lightweight and can be worn during the day while active, and at night while sleeping. It has been cleared by the FDA for treatment of chronic pain without a prescription. Users of the device have the option of using their smartphone to automatically track and personalize their pain therapy through the Quell Relief app, an updated version of which was released this week in the Apple app store.
Quell is an over-the-counter wearable pain relief device that utilizes proprietary non-invasive neurostimulation technology to provide pain relief. The device is designed for people with conditions such as diabetic nerve pain, low back and leg pain, fibromyalgia, and pain associated with osteoarthritis. The advanced wearable device is lightweight and can be worn during the day while active, and at night while sleeping. It has been cleared by the FDA for treatment of chronic pain without a prescription. Users of the device have the option of using their smartphone to automatically track and personalize their pain therapy through the Quell Relief app, an updated version of which was released this week in the Apple app store.
Shai N. Gozani, M.D., Ph.D., President and Chief Executive Officer of NeuroMetrix, stated:
“Initiating shipments of Quell represents a significant milestone for NeuroMetrix. This is an important step in completing our transformation from a traditional medical device company to one focused on the growing consumer healthcare market. Our employees have worked very hard to achieve this goal, and we are justifiably proud of this important achievement. We now look forward to growing Quell into a large and successful consumer healthcare brand.”
Have a crowdfunding offering you'd like to share? Submit an offering for consideration using our Submit a Tip form and we may share it on our site!