 Injeq Oy (Ltd), a Finland based MedTech company, has announced an equity crowdfunding round on Invesdor. Injeq will use the funding to bring their smart Injeq IQ-Needle to the European medical device markets. Think Spinal Tap, but not the movie. For medical professionals, however, the upcoming smart needle is predicted to make a permanent change for the better.
Injeq Oy (Ltd), a Finland based MedTech company, has announced an equity crowdfunding round on Invesdor. Injeq will use the funding to bring their smart Injeq IQ-Needle to the European medical device markets. Think Spinal Tap, but not the movie. For medical professionals, however, the upcoming smart needle is predicted to make a permanent change for the better.
“Many of our previous owners are physicians or dentists by profession which to us has always enforced that we are solving a real and worthwhile problem. Solving this problem also establishes a base for business and we are happy to now offer a larger crowd an opportunity to join us in the next phase of bringing the smart needle to everyday healthcare,” says Kai Kronström, CEO.
The lumbar puncture or “spinal tap” is said to be a rare procedure but it is not uncommon in hospitals. In the treatment of acute lymphocytic leukemia (ALL), which is the most common cancer type diagnosed in children, the medication will be injected in the spinal channel during a lumbar puncture. The treatment for ALL usually lasts for 30 months, during which time around 20 punctures are performed on the patient. Each puncture is a demanding procedure, because the tip of the needle must be inserted precisely into the spinal canal. If the needle is inserted even slightly too far, it can cause bleeding, which would allow cancer cells to enter the spinal canal, lessening the patient’s chances of recovery.
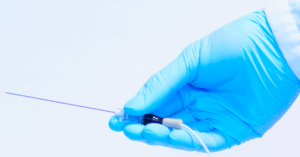 The tissue identification functionality of Injeq IQ-Needle is described as introducing a new standard level for both security and success, because it immediately alerts as the needle tip reaches its target; the spinal fluid.
The tissue identification functionality of Injeq IQ-Needle is described as introducing a new standard level for both security and success, because it immediately alerts as the needle tip reaches its target; the spinal fluid.
The company reports that the Injeq smart needle products have been used in clinical patient trials and the positive performance has been reported in the Journal of Clinical Monitoring and Computing. A lumbar puncture study with pediatric patients started in 2016 and is today proceeding at two university hospitals in Tampere and Turku.
New funding will allow the company to keep pace and to expand clinical studies to leading hospitals in Sweden, Norway and Denmark during the fall of 2017. Production is essential to the completion of final tests required for regulatory approval and the supply of needles both for research and commercial use. The initial market focus is in European Children’s Hospitals. Commercial deliveries are expected to take place during the first half of 2018.
“We are now in a situation where the design has been completed and we are ready to start production very quickly and make a first market entry in children’s hospitals. We have a unique product fulfilling an unmet clinical need,” says Kai Kronström, CEO.
The funding on Invesdor has topped the minimum €250,000 goal. The company is offering 3.84% to 11.64% equity in the business. Pre-money valuation is pegged at €6.26 million.
Have a crowdfunding offering you'd like to share? Submit an offering for consideration using our Submit a Tip form and we may share it on our site!

