Aromics, a privately-owned development stage pharmaceutical company founded in 2005, is based in Barcelona, Spain and focused on developing novel first-in-class therapies for the treatment of human diseases. The company has received the seal of Innovative SME from MINECO and achieved different R&D awards from international, European, national and local public organizations including the European Union, NCI, NIH, ICEX, CDTI, ENISA, ACC1Ó and MINECO. The company’s primary foci are oncology and infectious diseases.
 Since launching its crowdfunding campaign on Capital Cell, Aromics has raised just over 95,020€ toward its 300,000€ goal for 6.19% equity offered. Cofounded by CEO Dr. Carme Plasencia, CFO and Chairman of the Board Narcís Clavell and Immaculada Dalmau, Aromics is currently valued at 4,550,000€. A team of international advisers, associated partners and coworkers is already in place.
Since launching its crowdfunding campaign on Capital Cell, Aromics has raised just over 95,020€ toward its 300,000€ goal for 6.19% equity offered. Cofounded by CEO Dr. Carme Plasencia, CFO and Chairman of the Board Narcís Clavell and Immaculada Dalmau, Aromics is currently valued at 4,550,000€. A team of international advisers, associated partners and coworkers is already in place.
Aromics’ most advanced antitumor drug candidate is NAX035, a first-in-class, selective and potent compound for the treatment of cancer. The novel drug is currently completing regulatory preclinics compelling all the data required by the European Medicine Agency, seeking the approval for conducting Phase IB/II (safety and efficacy) clinical testing in patients.
“Malignant mesothelioma has been selected as first indication for NAX035 to proceed into clinics due to the potency exhibited by the compound in this tumor type including both, pleural and peritoneal cancers,” according to the campaign. “If effective, NAX035 will suppose a clear advance in the treatment of this rare cancer. Malignant mesothelioma is an aggressive tumor arising in the lining cells (mesothelium) of the pleural and peritoneal cavities, as well as in the pericardium and the tunica vaginalis. With a natural history of 7 to 9 months if untreated and less than five per cent 5-years survivors, is a bad prognosis tumor representing currently 0,3% of total cancer deaths.”
Why invest?
“Current treatment includes surgery, radiotherapy and chemotherapy. Surgery is the most effective option, although unfortunately aproximately 85-90% of patients are diagnosed at advanced stage where the disease is inoperable, and thus the treatment palliative. Survival time is of 6-12 months with a response rate to chemotherapy in advanced stage that do not exceed 50%. For this reason, we are looking for a new therapeutic strategy that substitutes the current treatment,” indicated Aromics. “The new agents in development include targeted therapies, monoclonal antibodies and immunoncologicals. None of them has exhibited yet superiority versus the standard of care. It is mandatory to find new and efficient treatments for the tumor, both as first and second-line therapy.”
 NAX035, a small molecule that has shown efficacy in front a variety of tumors of different origins including the most prevalent types of malignant mesothelioma (pleural and peritoneal) both, in vitro (in tumor cell lines derived from human sensitive and resistant tumors to pemetrexed-cisplatin therapy) and in vivo (in experimental xenograft mice) when it is administered orally or intravenously, exhibited an appropiate safety and tolerability profile in experimental animals. The compound is easy to manufacture at an affordable cost, which is important for future market penetration, said the company.
NAX035, a small molecule that has shown efficacy in front a variety of tumors of different origins including the most prevalent types of malignant mesothelioma (pleural and peritoneal) both, in vitro (in tumor cell lines derived from human sensitive and resistant tumors to pemetrexed-cisplatin therapy) and in vivo (in experimental xenograft mice) when it is administered orally or intravenously, exhibited an appropiate safety and tolerability profile in experimental animals. The compound is easy to manufacture at an affordable cost, which is important for future market penetration, said the company.
Crowdfund allocation is detailed below:
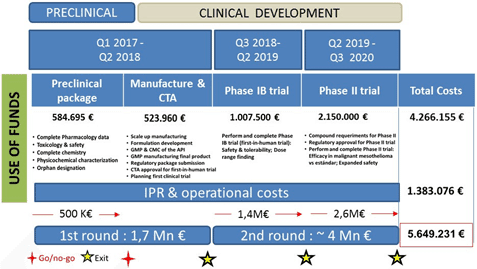
For additional detailed financial information and campaign updates, please click here.
___________________________________________________
Have a crowdfunding offering you'd like to share? Submit an offering for consideration using our Submit a Tip form and we may share it on our site!


 “Malignant mesothelioma has been selected as first indication for NAX035 to proceed into clinics due to the potency exhibited by the compound in this tumor type including both, pleural and peritoneal cancers,” according to the campaign. “If effective, NAX035 will suppose a clear advance in the treatment of this rare cancer. Malignant mesothelioma is an aggressive tumor arising in the lining cells (mesothelium) of the pleural and peritoneal cavities, as well as in the pericardium and the tunica vaginalis. With a natural history of 7 to 9 months if untreated and less than five per cent 5-years survivors, is a bad prognosis tumor representing currently 0,3% of total cancer deaths.”
“Malignant mesothelioma has been selected as first indication for NAX035 to proceed into clinics due to the potency exhibited by the compound in this tumor type including both, pleural and peritoneal cancers,” according to the campaign. “If effective, NAX035 will suppose a clear advance in the treatment of this rare cancer. Malignant mesothelioma is an aggressive tumor arising in the lining cells (mesothelium) of the pleural and peritoneal cavities, as well as in the pericardium and the tunica vaginalis. With a natural history of 7 to 9 months if untreated and less than five per cent 5-years survivors, is a bad prognosis tumor representing currently 0,3% of total cancer deaths.” “Current treatment includes surgery, radiotherapy and chemotherapy. Surgery is the most effective option, although unfortunately aproximately 85-90% of patients are diagnosed at advanced stage where the disease is inoperable, and thus the treatment palliative. Survival time is of 6-12 months with a response rate to chemotherapy in advanced stage that do not exceed 50%. For this reason, we are looking for a new therapeutic strategy that substitutes the current treatment,” indicated Aromics. “The new agents in development include targeted therapies, monoclonal antibodies and immunoncologicals. None of them has exhibited yet superiority versus the standard of care. It is mandatory to find new and efficient treatments for the tumor, both as first and second-line therapy.”
“Current treatment includes surgery, radiotherapy and chemotherapy. Surgery is the most effective option, although unfortunately aproximately 85-90% of patients are diagnosed at advanced stage where the disease is inoperable, and thus the treatment palliative. Survival time is of 6-12 months with a response rate to chemotherapy in advanced stage that do not exceed 50%. For this reason, we are looking for a new therapeutic strategy that substitutes the current treatment,” indicated Aromics. “The new agents in development include targeted therapies, monoclonal antibodies and immunoncologicals. None of them has exhibited yet superiority versus the standard of care. It is mandatory to find new and efficient treatments for the tumor, both as first and second-line therapy.”